 Semimetals exhibit properties intermediate between those of metals and nonmetals. The elements can be broadly divided into metals, nonmetals, and semimetals. Hydrogen, nitrogen, oxygen, fluorine, and chlorine. Some of the groups have widely-used common names, including the alkali metals (Group 1) and the alkaline earth metals (Group 2) on the far left, and the halogens (Group 17) and the noble gases (Group 18) on the far right. Since hydrogen is the first element and sometimes acts like the alkali metals, it is found in the left uppermost corner of the periodic table. Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 1–18 from left to right) the seven horizontal rows are called periods. This article considers the origin of the elements and their abundances throughout the universe. Elements are the fundamental materials of which all matter is composed. It arranges of the elements in order of increasing atomic number. chemical element, any substance that cannot be decomposed into simpler substances by ordinary chemical processes. The periodic table is used as a predictive tool. As expected, semimetals exhibit properties intermediate between metals and nonmetals. 
Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Heavy metals (HMs) are defined as those elements having an atomic number greater than 20 and atomic density above 5 g cm 3 and must exhibit the properties of. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. The vast majority of the known elements are metals. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. The Metallic elements table in this section categorises the elemental metals on the basis of their chemical properties into alkali and alkaline. The distinction between metals and nonmetals is one of the most fundamental in chemistry. Categories described in the subsections below include ferrous and non-ferrous metals brittle metals and refractory metals white metals heavy and light metals and base, noble, and precious metals. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. Unfortunately, there was a slightly different system in place in Europe.\) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line). The first two groups are 1A and 2A, while the last six groups are 3A through 8A. The traditional system used in the United States involves the use of the letters A and B. Metals are otherwise defined according to their position on the Periodic Table. 2.12) is a commonly used method of organizing the elements that provides useful information about the elements and their behavior. There are two different numbering systems that are commonly used to designate groups, and you should be familiar with both. The definition of metal: A substance with high electrical conductivity, luster, and malleability, which readily loses electrons to form positive ions ( cations ). These two rows are pulled out in order to make the table itself fit more easily onto a single page.Ī group is a vertical column of the periodic table, based on the organization of the outer shell electrons. Periods 6 and 7 have 32 elements, because the two bottom rows that are separate from the rest of the table belong to those periods. Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. A new period begins when a new principal energy level begins filling with electrons. There are seven periods in the periodic table, with each one beginning at the far left. 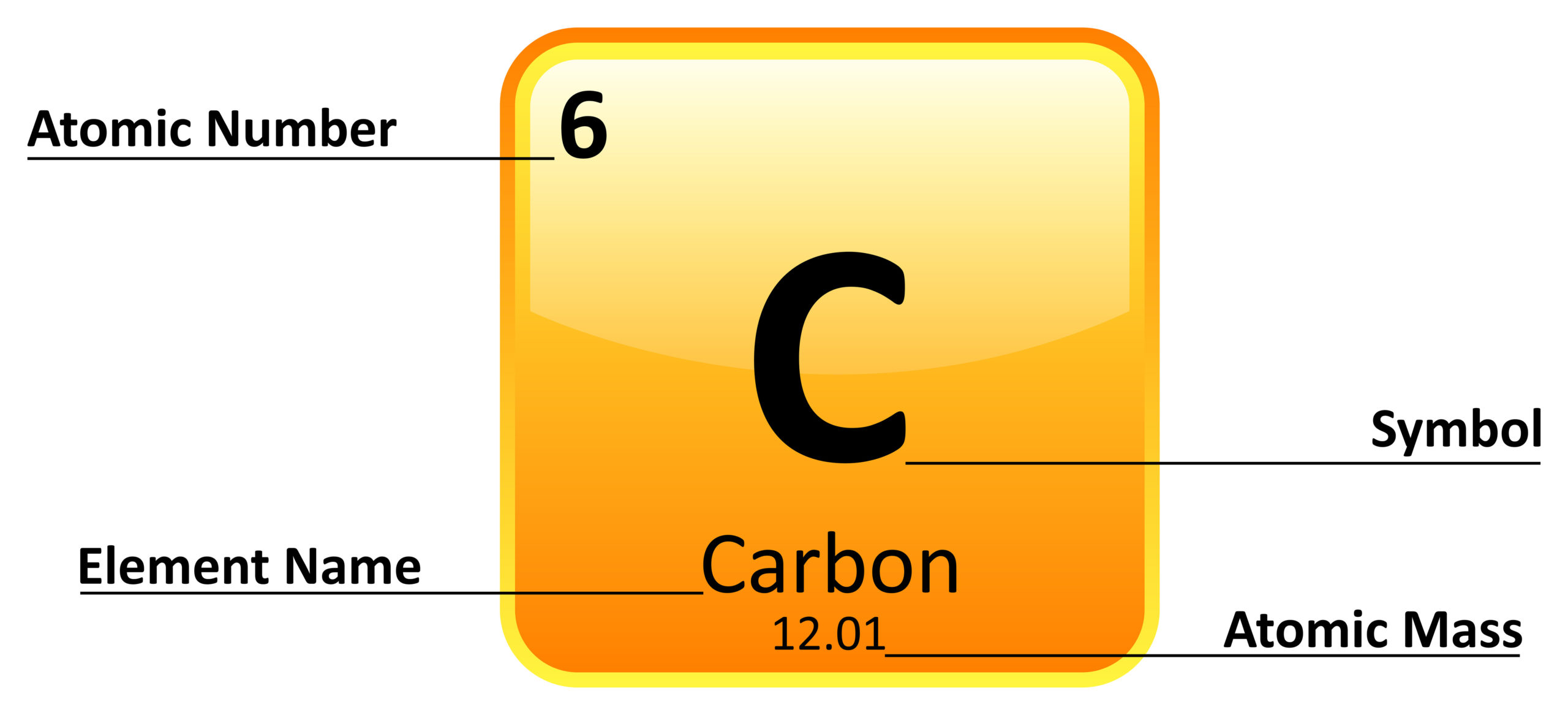
\) (Credit: User:Cepheus/Wikimedia Commons Source: (opens in new window) License: Public Domain)Ī period is a horizontal row of the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
